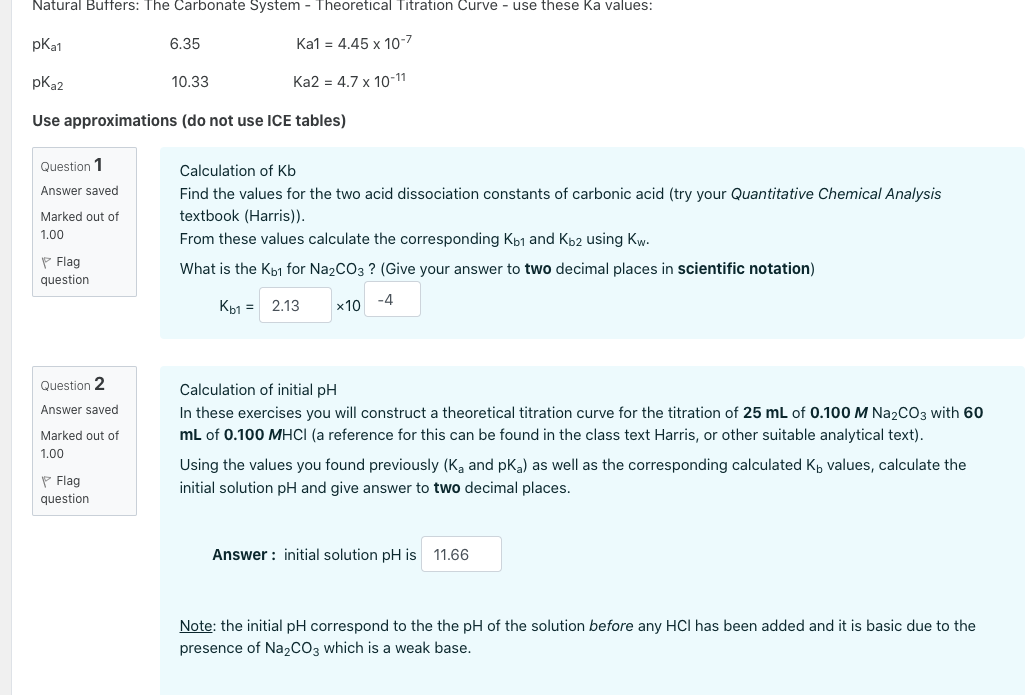
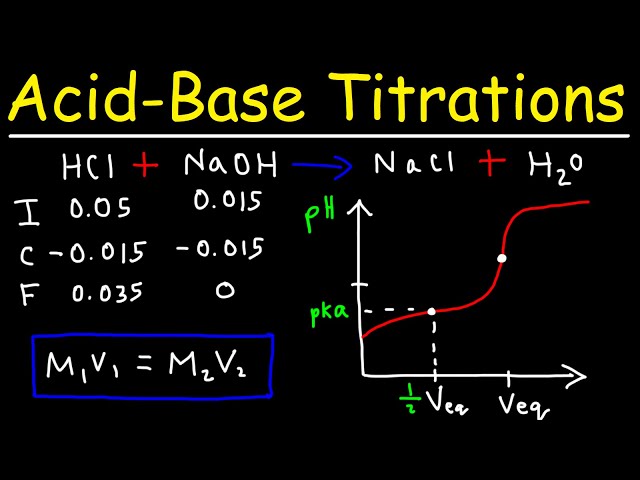
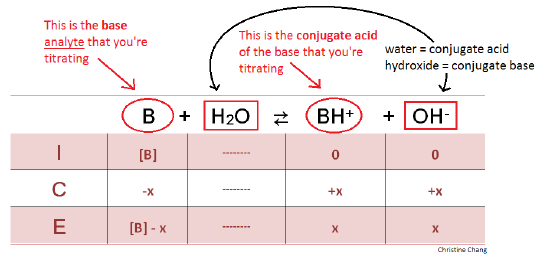
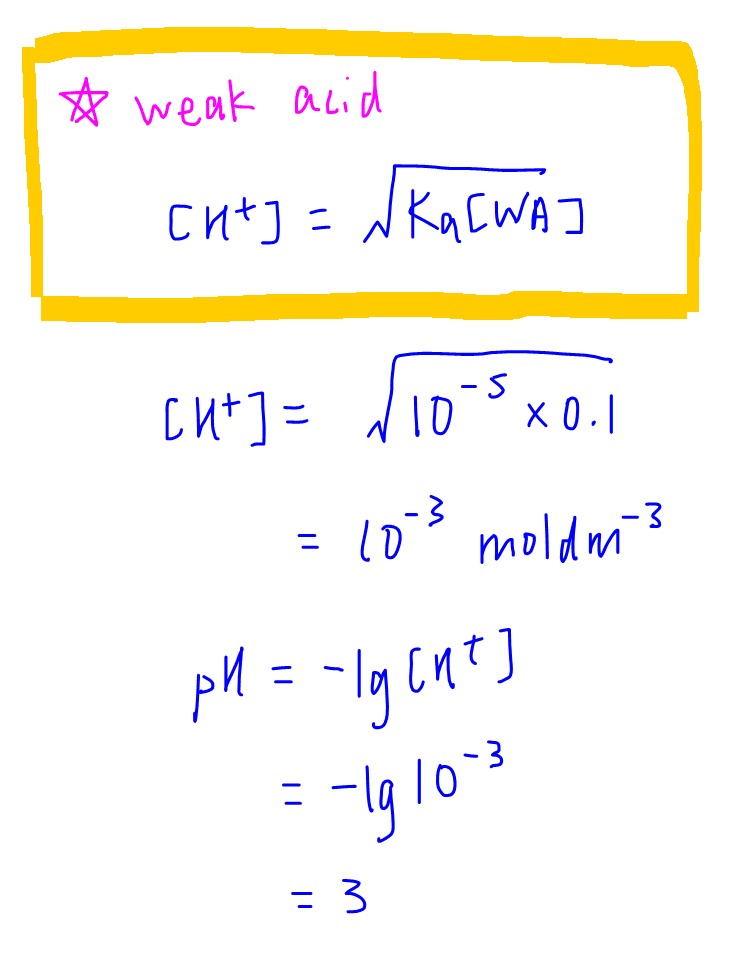In the titration of 50.0 mL of 0.10 M ammonia (K_b = 1.8 times 10^{-5}), calculate the pH: 1 ) Before titration begins 2 ) After addition of 20.0 mL of 0.10 M hydrochloric acid 3 ) After addition | Homework.Study.com

A level Indicator theory of acid-base titrations explained, theory pH curves & pKind, Indicator colour changes, Methyl orange, Bromophenol blue, Methyl red, Bromothymol blue, Phenol red, Thymol blue, Phenolphthalein GCE AS A2
![Calculate the pH at the equivalence point during the titration of 0.1M, 25 mL CH(3)COOH with 0.05M NaOH solution. [K(a)(CH(3)COOH) = 1.8 xx 10^(-5)] Calculate the pH at the equivalence point during the titration of 0.1M, 25 mL CH(3)COOH with 0.05M NaOH solution. [K(a)(CH(3)COOH) = 1.8 xx 10^(-5)]](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/18255401_web.png)
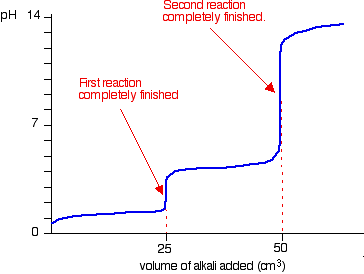
Calculate the pH at the equivalence point during the titration of 0.1M, 25 mL CH(3)COOH with 0.05M NaOH solution. [K(a)(CH(3)COOH) = 1.8 xx 10^(-5)]

University of South Carolina Board of Trustees Calculate the pH in the titration of mL of M HCl with M NaOH after a) 0.00 mL strong. - ppt download